Novel Foods
Last updated: 8 March 2023
On this page:
|
Summary
Note: This page considers the controls applied to “Novel Foods”. Originally this included genetically modified (GM) material used as food. However in 2003 separate EU controls were adopted. For details of the controls on GM food, please see the other page: GM Food Legislation in the EU. The following diagram illustrates the development of both the GM controls and those for “novel foods”:
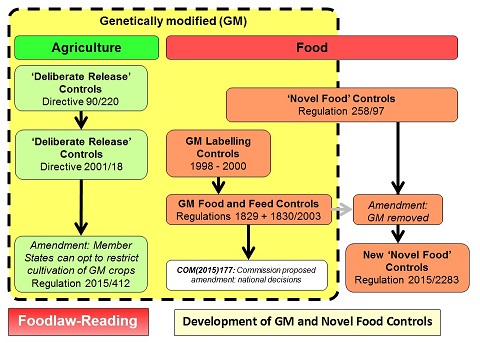
For a larger version of this figure, see: Diagram: GM and Novel Foods
Controls for “novel foods” were first proposed as part of the “Internal Market” programme in the early 1990s. Regulation 258/97 was eventually adopted in January 1997 and came into effect on the 15 May 1997. The date of 15 May 1997 has become important since any food or food ingredient commercially available in the EU prior to that date is not considered “novel”.
The Regulation applied to the following categories of foods and food ingredients (see Article 1(2)):
(a) foods and food ingredients containing or consisting of genetically modified organisms within the meaning of Directive 90/220/EEC;
(b) foods and food ingredients produced from, but not containing, genetically modified organisms;
(c) foods and food ingredients with a new or intentionally modified primary molecular structure;
(d) foods and food ingredients consisting of or isolated from micro-organisms, fungi or algae;
(e) foods and food ingredients consisting of or isolated from plants and food ingredients isolated from animals, except for foods and food ingredients obtained by traditional propagating or breeding practices and having a history of safe food use;
(f) foods and food ingredients to which has been applied a production process not currently used, where that process gives rise to significant changes in the composition or structure of the foods or food ingredients which affect their nutritional value, metabolism or level of undesirable substances.
The original regulation covered genetically modified foods and food ingredients - categories (a) and (b) above. These were however deleted when more detailed controls for GM foods were adopted in 2003.
Regulation 258/97 required “novel foods” to be subject to a risk assessment prior to their sale and use. This involved an initial assessment by a Member State and, in the event of an objection by another Member State to the initial assessment, a subsequent assessment by the European Food Safety Authority (EFSA). A significant number of novel foods were approved by this route and permitted by the adoption of Commission Regulations. Concerns were however raised by countries outside the EU that the Regulation created excessive barriers to foods which were common in other countries but which had not been commercially available within the EU prior to the 15 May 1997. The EU therefore agreed to amend and update the legislation.
The initial Commission proposal (COM(2002)0872) proved controversial in that it included procedures for the authorisation of nanotechnology and animal cloning in food production and, following a failed conciliation process, the proposal was not adopted. A subsequent proposal (COM(2013)0894), without the animal cloning element, was agreed and became Regulation 20015/2283. An extended definition of “novel food” is included - see Article 3(2). All risk assessments are now conducted by EFSA and a simplified route provides for the approval of “traditional foods from third countries”. All approved novel foods now appear in an official EU List published as Regulation 2017/2470 (as amended).
For the Commission's page on this topic, see: Novel Food. For an on-line “Novel Food Catalogue”, see the Commission's page: Novel Food Catalogue.
For details of the original EU controls and the process of adopting the current legislation, see: Novel Foods - Archive Page
Current EU Legislation
The following documents establish the updated controls on novel foods with effect from 1 January 2018
Regulation:
- Regulation (EU) 2015/2283 of the European Parliament and of the Council of 25 November 2015 on novel foods, amending Regulation (EU) No 1169/2011 of the European Parliament and of the Council and repealing Regulation (EC) No 258/97 of the European Parliament and of the Council and Commission Regulation (EC) No 1852/2001 (OJ L327, 11.12.2015, page 1)
- Amendment:
- Regulation (EU) 2019/1381 of the European Parliament and of the Council of 20 June 2019 on the transparency and sustainability of the EU risk assessment in the food chain and amending Regulations (EC) No 178/2002, (EC) No 1829/2003, (EC) No 1831/2003, (EC) No 2065/2003, (EC) No 1935/2004, (EC) No 1331/2008, (EC) No 1107/2009, (EU) 2015/2283 and Directive 2001/18/EC (OJ L231, 6.9.2019, page 1)
Administrative and Scientific Requirements:
- Commission Implementing Regulation (EU) 2017/2468 of 20 December 2017 laying down administrative and scientific requirements concerning traditional foods from third countries in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L351, 30.12.2017, page 55)
- Amendment:
- Commission Implementing Regulation (EU) 2020/1824 of 2 December 2020 amending Implementing Regulation (EU) 2017/2468 laying down administrative and scientific requirements concerning traditional foods from third countries in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L406, 3.12.2020, page 51)
- Commission Implementing Regulation (EU) 2017/2469 of 20 December 2017 laying down administrative and scientific requirements for applications referred to in Article 10 of Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L351, 30.12.2017, page 64)
- Amendment:
- Commission Implementing Regulation (EU) 2020/1772 of 26 November 2020 amending Implementing Regulation (EU) 2017/2469 laying down administrative and scientific requirements for applications referred to in Article 10 of Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L398, 27.11.2020, page 13)
- Commission Implementing Regulation (EU) 2018/456 of 19 March 2018 on the procedural steps of the consultation process for determination of novel food status in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L77, 20.3.2018, page 6)
Union List:
The following Regulation established the listing of approved novel foods. This Regulation has however been amended numerous times (approx.100) to include additional approved novel foods. Full details of the amending Regulation are not shown here but are given on a separate page - see: Novel Foods - Union List.
- Commission Implementing Regulation (EU) 2017/2470 of 20 December 2017 establishing the Union list of novel foods in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (OJ L351, 30.12.2017, page 72)
- For a complete corrected and republished new version of the Annex, see:
- Commission Implementing Regulation (EU) 2018/1023 of 23 July 2018 correcting Implementing Regulation (EU) 2017/2470 establishing the Union list of novel foods (OJ L187, 24.7.2018, page 1)
Consolidated Text: For a pdf consolidated text version of Regulation 2017/2470 with the full list in the Annex, see 2017/2470 Consolidated Text (published 29 August 2022). For any more recent amendments, see the Union List page mentioned above.
UK Legislation
Brexit: Prior to the IP Completion Day (31 December 2020), the legal requirements given in the EU Regulations listed above still applied to the UK. Since IP Completion Day, the EU Regulations above have been incorporated into UK legislation but with amendments to correct deficiencies. Information on this is given below. For more details of the process of incorporating EU legislation into UK law, see the separate page: UK Food Law: EU Legislation as Amended for the UK. Provisions for the enforcement of the controls (originally the EU Regulations but now as amended) have been provided in the UK Regulations listed below. For Northern Ireland, EU rules still apply.
1 January 2021: For interim guidance, see:
- Food Standards Agency: Novel foods guidance. See: https://www.food.gov.uk/business-guidance/novel-foods-guidance
- Food Standards Agency: Placing a regulated product on the market. See: https://www.food.gov.uk/business-guidance/placing-a-regulated-product-on-the-market
EU legislation with links to legislation.gov.uk: amended for application in the UK:
- Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel food … as amended by:
- Novel Food (Amendment) (EU Exit) Regulations 2019 (2019, No 702) as amended by:
- Food and Feed Hygiene and Safety (Miscellaneous Amendments etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1504)
- Food and Feed (Miscellaneous Amendments) Regulations 2022 (SI 2022, No. 1351)
- Commission Implementing Regulation (EU) 2017/2468 laying down administrative and scientific requirements concerning traditional foods from third countries … as amended by:
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702) as amended by:
- Food and Feed Hygiene and Safety (Miscellaneous Amendments etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1504)
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702) as amended by:
- Commission Implementing Regulation (EU) 2017/2469 laying down administrative and scientific requirements for applications referred to in Article 10 of Regulation (EU) 2015/228 as amended by:
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702 as amended by:
- Food and Feed Hygiene and Safety (Miscellaneous Amendments etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1504)
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702 as amended by:
- Commission Implementing Regulation (EU) 2017/2470 establishing the Union list of novel food …as amended by:
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702) as amended by:
- Food and Feed Hygiene and Safety (Miscellaneous Amendments etc.) (EU Exit) Regulations 2020 (SI 2020, No. 1504)
- Novel Food (Amendment) (EU Exit) Regulations 2019 (SI 2019, No 702) as amended by:
UK Legislation Post Brexit
- Novel Foods (Authorisations) and Smoke Flavourings (Modification of Authorisations) (England) Regulations 2022 SI 2022, No 560
- Novel Foods (Authorisations) and Smoke Flavourings (Modification of Authorisations) (Amendment) (England) Regulations 2022 SI 2022, No 619
Enforcement
Requirements for implementation and enforcement are provided separately for the four parts of the United Kingdom.
- England
- Novel Foods (England) Regulations 2018 (SI 2018, No. 154) as amended by
- Novel Food (Amendment) (EU Exit) Regulations (SI 2019, No 702)
- Wales
- Novel Foods (Wales) Regulations 2017 (SI 2017, No. 1103 (W.279)) as amended by
- Food (Miscellaneous Amendments) (Wales) (EU Exit) (No. 2) Regulations (SI 2019, No. 1046 (W.185))
- Novel Foods (Wales) Regulations 2017 (SI 2017, No. 1103 (W.279)) as amended by
- Scotland
- Novel Foods (Scotland) Regulations 2017 (SSI 2017, No. 415)
- Northern Ireland
- Novel Foods Regulations (Northern Ireland) 2017 (SRNI 2017, No. 233) as originally amended by
- Regulated Products (Amendment) (Northern Ireland) (EU Exit) Regulations (SI 2019, No. 849) [Note: revoked by SI 2020 No 1504]
Guidance
This page was first provided on 12 July 2010



